|
Step 1: Count the number of lone electron pairs on the central atom. Molecular geometry can be predicted using VSEPR by following a series of steps: In other words, the electrons will try to be as far apart as possible while still bonded to the central atom. The theory says that repulsion among the pairs of electrons on a central atom (whether bonding or non-bonding electron pairs) will control the geometry of the molecule. The valence shell electron-pair repulsion theory (abbreviated VSEPR) is commonly used to predict molecular geometry. Lewis structures are very useful in predicting the geometry of a molecule or ion.  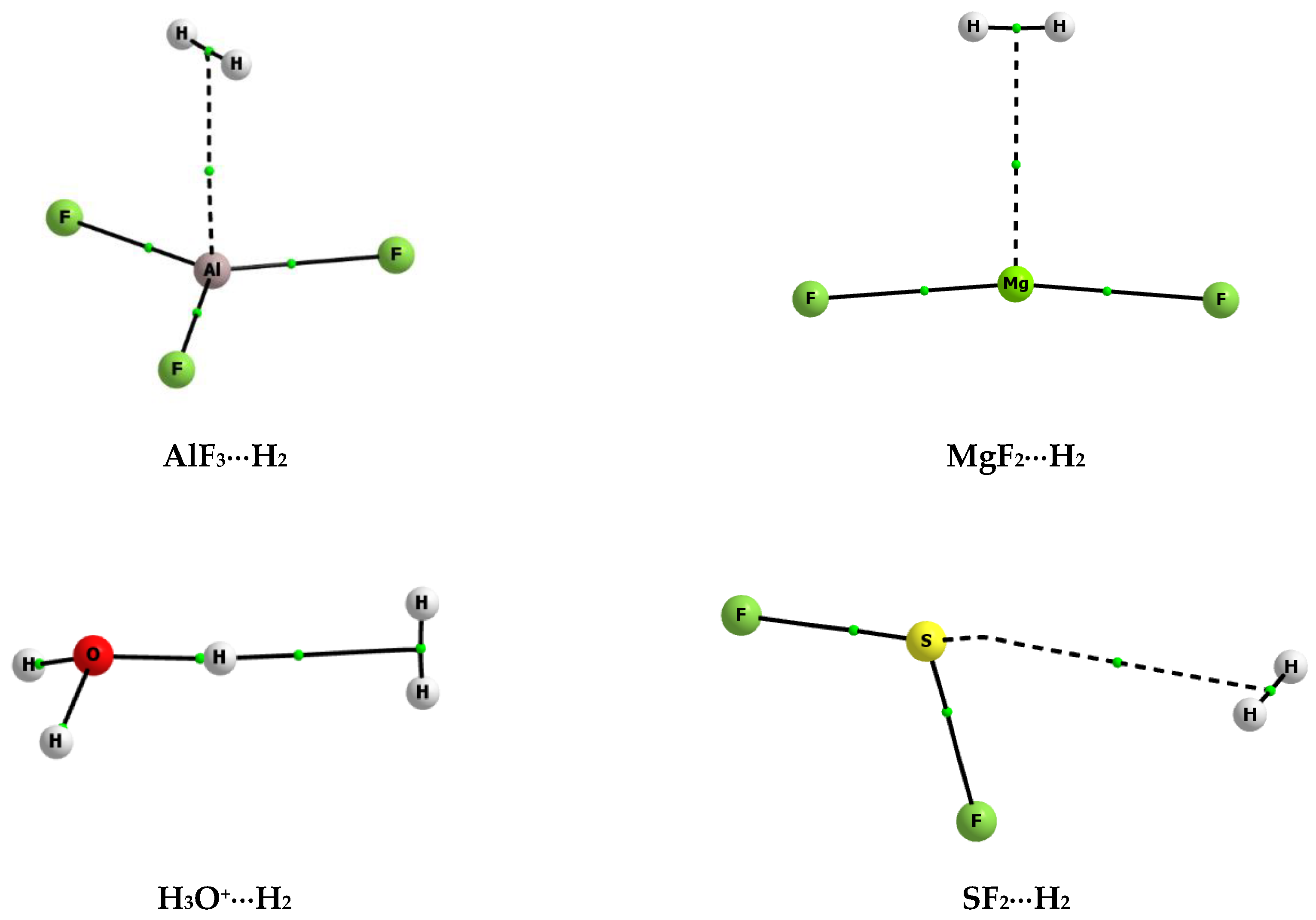 
Many of the physical and chemical properties of a molecule or ion are determined by its three-dimensional shape (or molecular geometry).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
